Woman Suffers Heart Failure After Taking A Weight Loss Product Bought From M'sian Seller
Three other women also developed palpitations and insomnia after taking similar weight loss products for several days.
A woman in Singapore is now suffering from severe heart failure after consuming a weight loss product containing an undeclared banned substance, which she had purchased from an online seller based in Malaysia
In an advisory issued on Monday, 3 June, the Singapore's Health Sciences Authority (HSA) said that the woman - who is in her 50s - developed an extremely fast heart rate (ventricular tachycardia) after consuming BB Body for three months, leading to a loss of consciousness that required resuscitation to save her life.
Now suffering from severe heart failure, she has been implanted with a defibrillator to regulate her heart rhythm and requires long-term heart failure medication.
BB Body is one of four products that were highlighted in HSA's advisory, which it said contained dangerous medicinal ingredients and had caused "serious adverse reactions" in consumers
The other products highlighted in the advisory are Bello Smaze and Choco Fit, both of which are marketed as weight loss products, as well as Seahorse Chop Du Zhong Ba Ji Wan, a health supplement which was found to contain a potent steroid.
Bello Smaze, which the HSA said contained sibutramine (an appetite suppressant), caused a woman in her 20s to develop palpitations and insomnia after taking it for four days. She also had suicidal thoughts in the short span of time.
The woman, who had purchased the product from a local seller to lose weight, noted that a friend who recommended the product to her also experienced similar symptoms.
In a separate case, another woman in her 30s developed palpitations after consuming Choco Fit - which also contained sibutramine - for two days.
According to Professor Chan Cheng Leng, group director of HSA's Health Products Regulation Group, sibutramine can cause mood disturbances, hallucinations, heart attacks, and strokes
"Do not fall for claims or promises of quick weight loss when purchasing health products, whether they are sold online or in retail shops," Chan advised consumers.
"Based on HSA’s enforcement operations, many of the weight loss products sold on e-commerce platforms without an established retail presence were found to contain the banned substance, sibutramine."HSA noted that BB Body, Bello Smaze, and Choco Fit were sold on various e-commerce and social media platforms, including Shopee, Instagram, and Facebook. They were often marketed as having "no side effects" or able deliver fast weight loss within days.
Meanwhile, a man in his 40s developed Cushing's syndrome after taking Seahorse Chop Du Zhong Ba Ji Wan, which he had purchased from a medical hall in Johor Bahru
The man had taken the "supplement" for two months to relieve his arthritis.
His condition, which the HSA noted is characterised by a 'moon face', thin limbs, thinning of skin, and easy bruising, was caused by dexamethasone, a potent steroid. HSA tests found that it also contained chlorpheniramine, an antihistamine for allergic reactions.
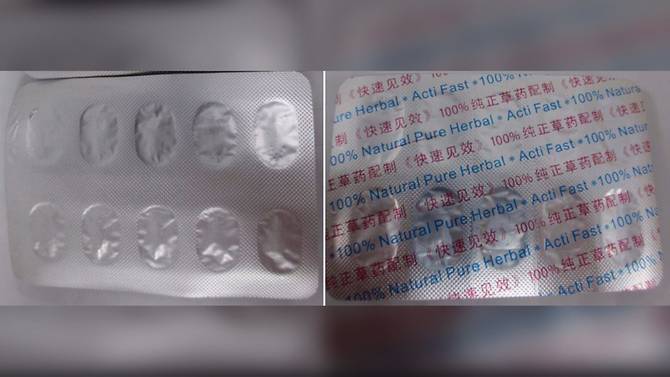
Another red flag raised by HSA was the blister strips found in one box, which carried a different product name and was labelled '100% Natural Pure Herbal * Acti Fast'. HSA later discovered that it contained frusemide, a diuretic used to eliminate water and salt from the body.
HSA cautioned consumers to stop taking the four aforementioned products immediately and advised them to consult a doctor immediately, especially if they had consumed the steroid-laden Seahorse Chop Du Zhong Ba Ji Wan
It also advised consumers to avoid purchasing health products from unfamiliar sources overseas, and exercise caution when buying such products online.
"You cannot be certain where and how these products were made. They could potentially be counterfeits or contain undeclared ingredients which can seriously harm your health", it said.
Sellers and suppliers are also ordered to stop selling the products immediately.
You can read HSA's full report here.